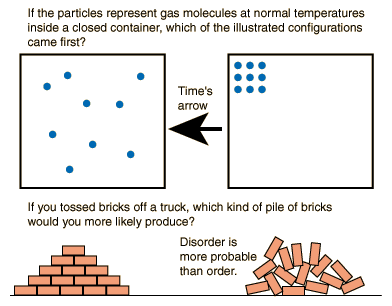
The rest heat is dumped in the cold sink, generating a lot of entropy. Some of this energy is converted to work, which has no effect on the entropy. Remember the steam engine from the eighteenth century? Energy leaves the hot source as heat, reducing its entropy (a sneeze in a loud street). This bring us back to Clausius’ statement from our previous blog. A sneeze in a quiet library will be much louder than the same sneeze in a busy street, although a similar amount of energy is released. To grasp this, I would like to use the analogy of the sneeze. Wherever order emerges, we must lift the curtain and see greater disorder being produced elsewhere. To resolve this paradox, you need to understand that change is not an island of activity in itself. In all seasons we see babies being born, cities built out of sand, wood and dust. If the state of everything we know is degenerating, even on a cosmic scale, why do we see so much ‘order’ in our everyday life? Come the spring, plants will grow and young leaves sprout out of trees. It is the rise in disorder of all the energy and matter in the universe. So, entropy is not something physical which is added to the universe. The greater the disorder, the greater the entropy. The molecules have turned from an orderly state (solid) into a disorderly state (gas).Īt the end of the nineteenth-century Austrian physicist Ludwig Boltzmann showed that entropy is a measure of disorder. This leaves more space in between the molecules and occupies a much larger volume. Turn up the heat and the liquid becomes a gas composed of molecules that fly on a collision course and bounce off each other. As the liquid flows, the atoms too start to get livelier. Molecules let go of the grid and start wobbling over each other. When the temperature rises the solid becomes a liquid. Each molecule fits neatly into the grid and – except for some freely floating electrons – nothing moves. Part three: the way the universe will end Disorder on a molecular scaleīehold the molecules of solid body, a huge wall tightly packed together. Part one: a simple definition Part two: birth of the Second Law What is the effect of this on our everyday lives? To better understand let’s zoom in on the molecular basis of matter. One of the rules about entropy is that it cannot decrease. Where q is the heat transferred in joules and ΔT is the change in temperature in Kelvin.What is Entropy? Part 3 the way the universe will endĮver wondered why heat never flows from cold to hot? Why we can never create a machine with 100% efficiency or how we know our past but not our future? It all has to do with an elusive concept called entropy. If there are 2 moles of reactant forming 1 mole of product, the number of states possible decreases and entropy decreases, likewise, if 1 mole of reactant forms 2 moles of product, the entropy increases.Įntropy can be calculated using the equation 
Example of increasing entropy: Ice melting in a warm room is a common example of increasing entropy. If a substance goes from a liquid to a solid, the molecules in the system occupy a lower number of states (think about the decrease of molecule activity in water when it freezes to ice) and therefore the entropy decreases if a substance goes from a solid to a liquid, the entropy increases.  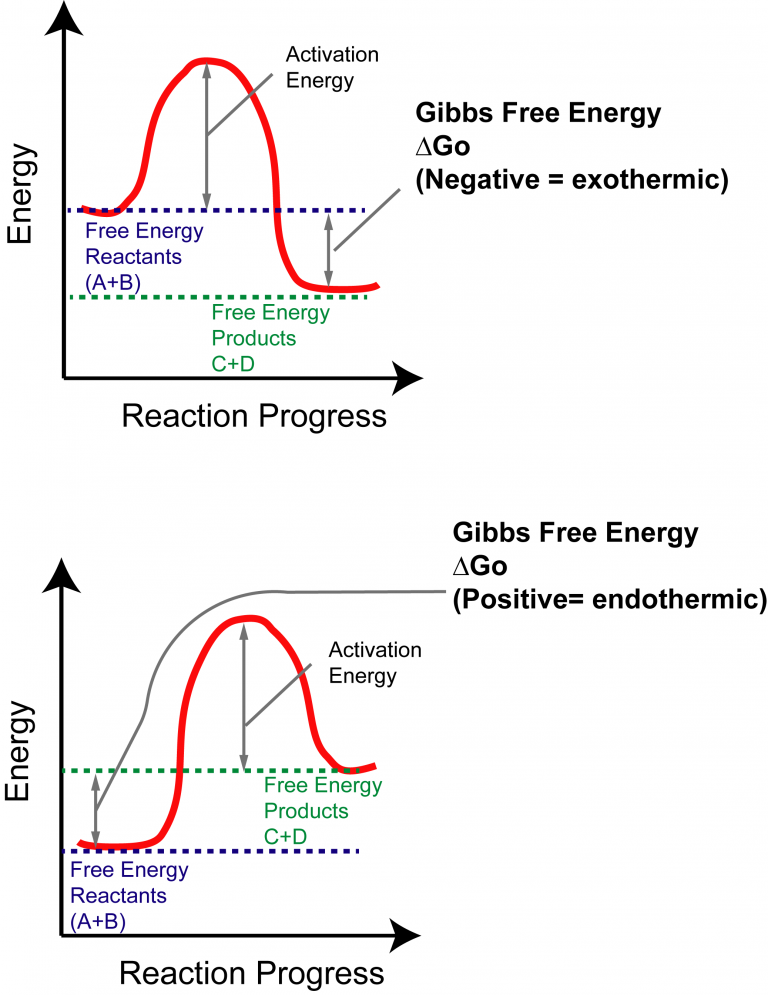
When considering whether reactions have an increase or decrease in entropy, two factors are important: The number of moles created and states of the reactants versus the products. Liquids can be considered the second most disordered, and solids can be considered the least disordered. Because molecules in gases are able to change both volume and shape, they can be considered the most disordered. We can think of the degrees of the number of different states the molecules in each can occupy. Solids, liquids, and gasses all have different degrees of disorder. The concept of disorder can best be described in terms of the states of matter. The unit of entropy is Joules per Kelvin or J/K.Įntropy is the measure of the disorder of a system and can is the energy of a system over its temperature, represented as J/K. The Second Law of Thermodynamics states that the entropy (or disorder) of a system is constantly increasing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
